Overview of Experiment:
Shelled organisms and organisms that create calcium carbonate skeletons are threatened by the drop in ocean pH (rise in ocean acidity) related to increased atmospheric carbon dioxide levels. Increasing acidity/lowered pH in ocean water not only causes the shells to dissolve, but it reduces the availability of carbonate ions – which
animals use to build their shells and skeletons.
Ca 2+ + CO3 2- -> CaCO3
Calcium + Carbonate -> Calcium Carbonate
You will observe the effect of an acidic environment on mussel shells through observations and measurements.
Hypothesis:
If e put mussel shlls in an acidic environment, then it will dissolve and weaken.
Material Checklist:
Student Pre- lab Questions:
1. How do organisms make their shells? What are shells made of?
They make their shells from water. The shells are of CaCO3.
2. What do you expect to happen to the shell in an acidic solution such as vinegar?
Their shells will dissolve or have some reaction to it in their shells.
3. What are sources of carbon dioxide and which of these sources are most likely to affect ocean pH?
Hydrothermal Vents.
Lab Procedure:
1. MATERIALS: Remove your two untreated shells from their bags.
2. MATERIALS: With a sharpie, label one shell “E’ for experimental and one shell “C” for control.
3. RECORDER: Using your data table record your group’s initial observations of the control and experimental shell characteristics.
4. MATERIALS: Find the mass of each of the shells.
5. RECORDER: Record the starting masses of both shells on your data table.
6. MEASURING: Pour 150 ml of vinegar into a 500 ml beaker and 150 ml of salt water into a second 500ml beaker.
7. TIMER: Set the timer for 30min.
8. MATERIALS: At the same time: Add the untreated, control shell “C” shell to salt water and the untreated, experimental shell “E” to the beaker of vinegar and start the timer.
9. RECORDER: In your data table, observe and record your group’s observations of what is happening to the shell while exposed to the vinegar over time. (at 0 minutes and 15 minutes)
Shelled organisms and organisms that create calcium carbonate skeletons are threatened by the drop in ocean pH (rise in ocean acidity) related to increased atmospheric carbon dioxide levels. Increasing acidity/lowered pH in ocean water not only causes the shells to dissolve, but it reduces the availability of carbonate ions – which
animals use to build their shells and skeletons.
Ca 2+ + CO3 2- -> CaCO3
Calcium + Carbonate -> Calcium Carbonate
You will observe the effect of an acidic environment on mussel shells through observations and measurements.
Hypothesis:
If e put mussel shlls in an acidic environment, then it will dissolve and weaken.
Material Checklist:
- (4) shells (2 untreated and 2 pre-treated in vinegar)
- (1) tweezer
- 150 mL vinegar
- (3) paper towels
- 150 mL salt water
- (1) sharpie
- (1) beaker
Student Pre- lab Questions:
1. How do organisms make their shells? What are shells made of?
They make their shells from water. The shells are of CaCO3.
2. What do you expect to happen to the shell in an acidic solution such as vinegar?
Their shells will dissolve or have some reaction to it in their shells.
3. What are sources of carbon dioxide and which of these sources are most likely to affect ocean pH?
Hydrothermal Vents.
Lab Procedure:
1. MATERIALS: Remove your two untreated shells from their bags.
2. MATERIALS: With a sharpie, label one shell “E’ for experimental and one shell “C” for control.
3. RECORDER: Using your data table record your group’s initial observations of the control and experimental shell characteristics.
4. MATERIALS: Find the mass of each of the shells.
5. RECORDER: Record the starting masses of both shells on your data table.
6. MEASURING: Pour 150 ml of vinegar into a 500 ml beaker and 150 ml of salt water into a second 500ml beaker.
7. TIMER: Set the timer for 30min.
8. MATERIALS: At the same time: Add the untreated, control shell “C” shell to salt water and the untreated, experimental shell “E” to the beaker of vinegar and start the timer.
9. RECORDER: In your data table, observe and record your group’s observations of what is happening to the shell while exposed to the vinegar over time. (at 0 minutes and 15 minutes)
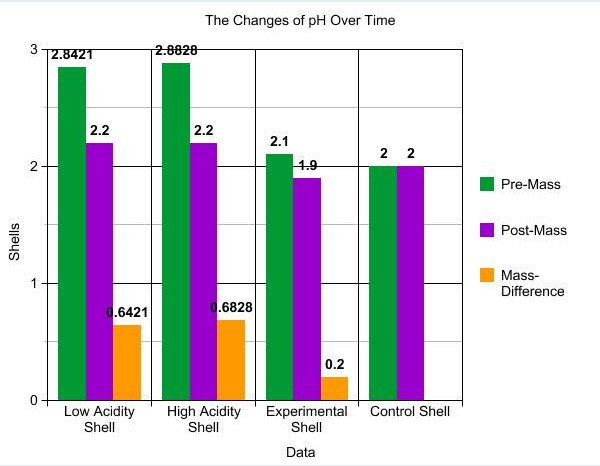
Shells Pre-Observations Post Observations Pre Mass Post Mass Mass Difference
L pre rough & less color "" 2.8421gr 2.2 gr 0.6421 gr
in the inside
H Pre More color, chipped "" 2.8828gr 2.2 gr 0.6828 gr
and damaged
E White in the inside , Is getting bubbles all over the 2.1 gr 1.9 gr 0.2gr
same texture as shell C shell lighter chipping
C Cracked, same texture Is cracked and grey , 2.0 gr 2.0 gr No difference.
as shell E Rough and black
L pre rough & less color "" 2.8421gr 2.2 gr 0.6421 gr
in the inside
H Pre More color, chipped "" 2.8828gr 2.2 gr 0.6828 gr
and damaged
E White in the inside , Is getting bubbles all over the 2.1 gr 1.9 gr 0.2gr
same texture as shell C shell lighter chipping
C Cracked, same texture Is cracked and grey , 2.0 gr 2.0 gr No difference.
as shell E Rough and black